Novartis' Ballet
Germán Müller – Business Development Manager
Exelon® should be featured in pharmaceutical marketing courses as one of the best success stories when it comes to life cycle management. In the following article, we tell the reasons
The Stage
In spite of there is currently no treatment that can cure dementia or reverse its progressive course, there are numerous interventions available to improve the lives of people with dementia, their care givers and families.
This condition has been one of the main focuses targeted by the pharmaceutical industry in recent decades within the framework of the Central Nervous System (CNS).
In addition, world population growth and the constant increase in life expectancy show that dementia will continue to be one of the most serious health problems globally, as explained in detail here.
In this article we will focus on the strategy carried out by Novartis in reference to the life cycle management of Exelon®, from the launch of oral Rivastigmine in April 2000 and until the arrival of generic Rivastigmine patches, which occurred between 2015 and 2019.
Timing and rhythm
Rivastigmine as a molecule was patented in 1985 and came into medical use in 1997. The molecule was developed by Marta Weinstock-Rosin of the Department of Pharmacology at the Hebrew University of Jerusalem and sold to Novartis by Yissum (the technology transfer company of the university) for commercial development.
Subsequently, in the 21st of April of 2000, Novartis received the approval from the FDA for the Exelon® Rivastigmine tartrate capsules, indicated for the treatment of mild to moderate dementia of the Alzheimer’s type or associated with Parkinson’s disease.
While the company was recovering its years of investment at the pace of flourishing sales, it was already focused on the next steps to anticipate the erosion of value that the market would go through at the unavoidable entry of generic products.
The cornerstone of this strategy was given by the development of a Rivastigmine patch, which would be supported by a large and solid clinical trial, which leads to the FDA approval of Exelon® Patch on July 6th, 2007. The constant delivery of Exelon® through the patch showed equivalent efficacy at the target dose to the highest doses of capsules but with three times fewer reports of nausea or vomiting. Thus, this product became the first and only transdermal therapy approved to treat all stages of Alzheimer’s.
Given this approval and in light of the advantages offered by the transdermal patch, Novartis then focused on the migration of patients who were being treated with the oral presentation of Exelon® (as well as some substitute molecules, such as Donepezil and Memantine) towards the recently approved transdermal patch.
By 2010, three out of four people treated in the United States with Rivastigmine were through the transdermal dosage form. This means that in less than three years, Novartis had already managed to migrate 75% of Rivastigmine sales to the new pharmaceutical form, still being the only company that offered both alternatives.

Finally, on the 1st of July of 2010 Sun Pharma announced the launch of generic Exelon®, Rivastigmine tartrate capsules for the US market, which was previously approved by the FDA as an ANDA with a paragraph IV certification by October 2007.
On the same day, at Princeton, New Jersey; Sandoz announced the introduction of rivastigmine tartrate capsules, an authorized generic equivalent of the Novartis treatment Exelon® capsules.
This was followed a few days later by Watson Pharmaceuticals, reporting that, under a settlement with Novartis commenced the commercialization of a generic version of Exelon®. Situation that will be repeated by Dr. Reddy´s and Alembic/Breckenridge in 2011 and 2012 accordingly.
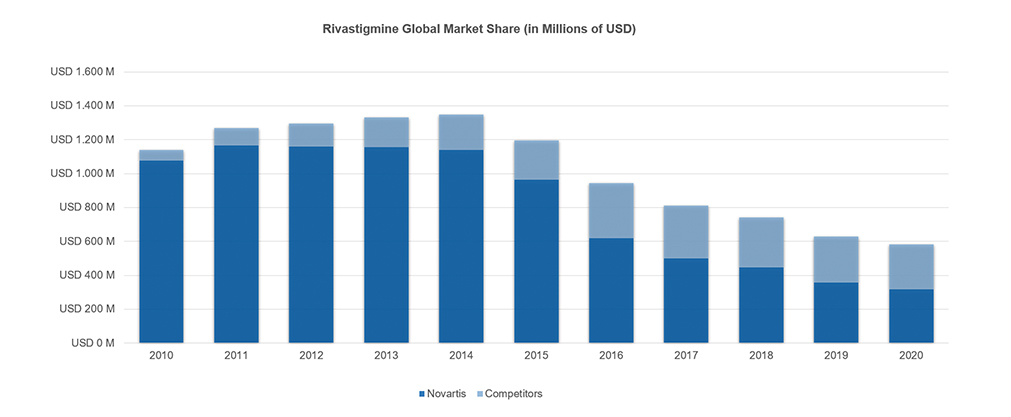
Finally, on August 31st, 2015, Alvogen launched the first generic of a Rivastigmine patch to the US market on what represented a postponed turn on the course of this harmonious masterpiece of Novartis. Which at that time held more than 80% of the global Rivastigmine market, which would it represented close to one billion dollars in sales, with half of these occurring in the United States where its market share was even larger (90%).
That was the beginning of a major penetration of generic products into the Rivastigmine market, which would be complemented between 2018 and 2019 with the entry of new generic patches (Mylan, Amneal, Zydus, Breckenridge), burying the total sales for this molecule to a fifth of its value over the course of 5 years. Thus, sales shrunk from the historical maximum of sales achieved in the US in 2014 with almost USD 650, to a turnover of around USD 120 M for the year 2020, of which Novartis still owned almost 60%.
Main Actors
There is a multiplicity of factors that allowed Novartis to perpetuate over time its position as the dominant leader in the Rivastigmine market across different pharmaceutical forms, but there are three that stand out from the rest since they turned out to be decisive.
The first of these is probably the most evident in the pharmaceutical industry, since it is the essential patent protection, an elementary factor in a sector of the economy that involves extended periods of research with a strong investment in R&D.
The first patent related to Rivastigmine was granted in 1990 to its inventor, which was in force until 2012, after obtaining a 5-year extension. It was succeeded by a series of patents that protected the transdermal form and the use of antioxidants in this pharmaceutical form. The first patent of this second group expired in February 2014, which allowed the entry of certain generic patches that were able to circumscribe the patents still in force, whose expiration occurred in January 2019.
The second essential factor was the robust clinical study carried out by Novartis to benefit an swift and efficient transition of patients from oral Rivastigmine to the transdermal presentation, based fundamentally on the advantages offered by the patch.
The efficacy of, and caregiver preference for, the rivastigmine transdermal patch versus rivastigmine capsules were assessed in the large (n = 1195), 24-week, randomized, double-blind, double-dummy, placebo- and active comparator controlled, multinational IDEAL (Investigation of transDermal Exelon® in ALzheimers disease) trial.
Patients receiving 24 weeks of treatment with rivastigmine transdermal patch in this study, experienced significant improvement in global and cognitive function, with that improvement being noninferior to that observed with rivastigmine capsules, but with one-third fewer reports of nausea and vomiting. Moreover, most (>70%) caregivers preferred the patch over the capsules, mainly because of the ease of following the schedule and the ease of use.
Finally, the last central element that allowed Novartis to make out of Exelon® a success business case lies in the price of the product, since at the time of making the transition there was no significant impact for the various parties involved in dealing with the costs of this therapy. As mentioned, when Novartis’ launched the patch there were still no competitors in the oral form, which would have represented some price pressure that had made the transition to a patch, typically more expensive than oral forms, more complex.
The evaluation for the cost of the medication was well addressed by Novartis on the mentioned study. A pharmacoeconomic analysis conducted from the perspective of the UK National Health Service (NHS) and based on MMSE scores from IDEAL estimated that, relative to best supportive care, the incremental cost per quality-adjusted life-year (QALY) gained with rivastigmine patch was £10.579 compared with £13.042 with rivastigmine capsules (year of costing 2008/9). Similar results were observed when the base case analysis was based on the MMSE as well as the ADL scores, with estimated incremental costs per QALY of £9.114 for rivastigmine patch and £13.758 for rivastigmine capsules versus best supportive care. Although additional pharmacoeconomic data are required, these data suggested that rivastigmine patch may provide a cost effective treatment option for patients with Alzheimer’s disease.
A final comment with regards to the influencing components in this analysis has to do with the intentional omission of the timing in the execution of the strategy as it is considered obvious, since any delay that may have occurred in the development of the patch, the clinical studies, the approvals from the health authorities or the migration of patients from the tablet to the patch (among others), would undoubtedly have generated some unbalance in this perfectly synchronized ballet piece.
The evolution
Almost 15 years after the launch of Exelon® transdermal, Luye Pharma Group announced that marketing approval for its innovative formulation Rivastigmine Multi-Day Transdermal Patch (Rivastigmine MD) has been granted by the UK’s Medicines and Healthcare Products Regulatory Agency, for the treatment of mild to moderate dementia associated with Alzheimer’s disease. The drug has previously received marketing authorization for several EU countries.
Rivastigmine MD is a twice-weekly innovative patch formulation for the treatment of mild to moderate dementia associated with Alzheimer’s disease, and introduce an alternative to the existing options: daily tablets and patches.
Amarin is currently commercializing its improved daily patch, as it is 24% reduced in size and drug content than the Exelon®, hence it is 131% more efficient. Additionally, our formulation has 50% longer shelf life (24 month of Exelon® vs 36 months for Amarin´s Patch).
In order to continue with the evolution of the product and bringing innovation to the transdermal field, at Amarin we are also working on a one-a-week rivastigmine patch that would provide benefits to both the patient and the families of affected individuals and their caregivers.
References:
- Nagy B, Brennan A, Brandtmuller A, et al. Assessing the cost-effectiveness of the rivastigmine transdermal patch for Alzheimer’s disease in the UK using MMSE- and ADLbased models. Int J Geriatr Psychiatry 2011 May; 26 (5): 483-94
- Winblad B, Cummings J, Andreasen N, et al. A six-month double-blind, randomized, placebo-controlled study of a transdermal patch in Alzheimer’s disease-rivastigmine patch versus capsule. Int J Geriatr Psychiatry 2007 May; 22 (5): 456-67
- Rivastigmine Transdermal Patch A Review of its Use in the Management of Dementia of the Alzheimer’s Type Sohita Dhillon Adis, a Wolters Kluwer business, Auckland, New Zealand
- Therapies for Alzheimer’s disease – Irena Melnikova – www.nature.com/reviews/drugdisc / May 2007 (VOLUME 6)
- https://www.fiercebiotech.com/biotech/fda-approves-novartis-alzheimer-s-patch
- https://www.luye.com/en/info.php?id=323
Subscribe to
#AmarinNews
Partner with us!
We offer expertise and experience, together with flexibility and the ability to adapt to your needs.
- info@amarintech.com.ar
- +54 11 4588-6500
- Sanchez 2045 (C1416BQG), Buenos Aires, Argentina.



